Abstract
Haploidentical hematopoietic cell transplant (HaploHCT) with post-transplant cyclophosphamide (PTCy) has expanded the donor pool and made HCT available to patients without a matched donor. Cytokine release syndrome (CRS) is a syndrome of systemic inflammation with massive cytokine release, originally described after chimeric antigen receptor T cell (CAR T) therapy, is a frequent complication early after HaploHCT. Severe CRS after HaploHCT has been associated with higher non-relapse mortality (NRM) and worse overall survival (OS). Currently, no standard guidelines exist for CRS management in HaploHCT patients other than general supportive measures. Here, we described the impact of tocilizumab, an interleukin-6 receptor inhibitor used to manage CRS in CAR T therapy setting, on clinical outcomes in patients who experienced CRS after HaploHCT.
We retrospectively identified a case series of 120 patients who experienced CRS after undergoing HaploHCT with PTCy-based GvHD prophylaxis at City of Hope from 06/2019 to 06/2021; of whom 11 patients received tocilizumab for CRS management. We matched these patients in a 1:2 ratio with 21 patients with CRS post-Haplo-HCT who did not receive tocilizumab (based on age, primary diagnosis, conditioning regimen, and CRS grading), then analyzed and compared OS, disease-free survival (DFS), relapse rate (RR), NRM, cumulative incidence (CIN) of grade II-IV and III-IV acute GvHD, and chronic GvHD between the two groups by univariate analysis. P-values were 2 sided at a significance level of 0.05.
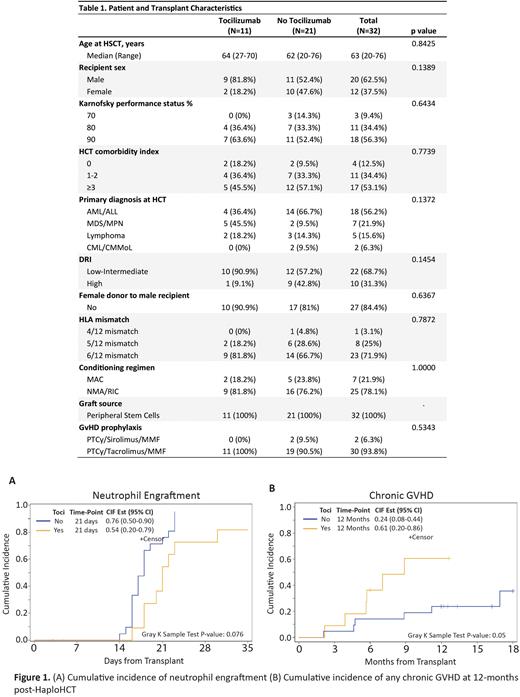
Table 1 summarize patient and transplant characteristics. Median age of patients in tocilizumab (TOCI) and no-tocilizumab (NO-TOCI) cohorts were 64 years (range: 27-70) and 62 years (range: 20-76). HLA 6/12 mismatch was the most common in both cohorts (81.8% in TOCI and 66.7% in NO-TOCI). All patients received mobilized peripheral blood stem cells (PBSC) as graft source. Majority of patients received non-myeloablative or reduced intensity conditioning regimens (81.8% in TOCI and 76.2% in NO-TOCI). Grade 2 CRS was most common in both cohorts (72.2% in TOCI and 71.4% in NO-TOCI); and grade 3-4 CRS was documented in 1 patient (9.1%) in TOCI and 2 patients (9.5%) in NO-TOCI cohort. Tocilizumab doses were 4 mg/kg (n=2), or 8 mg/kg (n=9). All patients but one (90.9%) received one single dose of tocilizumab and 1 patient received 2 doses of tocilizumab at 8 mg/kg/dose. All tocilizumab doses were administered either on day +3 (45%) or day +4 (55%) post-HCT (the patient with 2 doses was counted towards day +3).
No high-grade tocilizumab-related side effects were documented in those 11 patients. Day 28 CIN of neutrophil engraftment was lower in TOCI compared to NO-TOCI patients (54.5% vs 76.2%, 95% CI: 21-79 and 50-90%, P=0.07 - Figure 1a). At 12 months post HaploHCT, survival outcomes were similar with NRM of 18.2 and 19% (95% CI: 3-46% and 6-38%, P=0.92); and RR of 19.5 and 14.3% (95% CI: 3-49% and 3-33%, P=0.78) in TOCI and NO-TOCI cohorts, respectively. DFS and OS were not affected by tocilizumab administration (P=0.90 and P=0.98; respectively). Three patients in TOCI and 7 patients in NO-TOCI cohort died; their causes of death were not directly related to CRS. While CIN of day 100 grade II-IV (P=0.97) and grade III-IV acute GvHD (P=0.47) were not affected by tocilizumab administration, CIN of chronic GvHD was significantly higher in TOCI compared to NO-TOCI (60.6 vs 23.8%, 95% CI: 19-86% and 8-44%, P=0.05 - Figure 1b).
In conclusion, our results indicates that tocilizumab used for CRS management after in HaploHCT setting with PTCy-based GvHD prophylaxis was well tolerated and had no effect on survival outcomes. However, our study showed a trend towards delayed neutrophil engraftment and a statistically significant higher incidence of chronic GvHD in patients who received tocilizumab. It is possible that tocilizumab may interfere with T cell expansion, which could impact PTCy-mediated elimination of alloreactive T cell clones and/or affect regulatory T cells function; both mechanisms could lead to higher incidence of chronic GvHD. Using this approach to mitigate CRS risk should be reserved to patients with high grade CRS where mortality is high.
Disclosures
Ball:Oncovalent: Membership on an entity's Board of Directors or advisory committees. Koller:Treadwell Therapeutics: Other: Safety Review Committee; Takeda: Speakers Bureau; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Salhotra:Kadmon: Other: Advisory board meeting ; Orca Bio: Research Funding; BMS: Research Funding. Aribi:SeaGen: Consultancy. Artz:Abbvie: Honoraria; Magenta: Honoraria. Aldoss:Autolus Limited: Consultancy; Agios: Consultancy, Honoraria; AbbVie: Consultancy, Research Funding; Amgen: Consultancy; Jazz Pharmaceuticals: Honoraria, Speakers Bureau; Kite: Consultancy. Ali:Abbvie: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees; Incyte Corporation: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Stein:Amgen: Speakers Bureau. Marcucci:Abbvie: Other: Speaker and advisory scientific board meetings; Agios: Other: Speaker and advisory scientific board meetings; Novartis: Other: Speaker and advisory scientific board meetings. Nakamura:BluebirdBio: Consultancy; Omeros: Consultancy; Sanofi: Consultancy; Magenta Therapeutics: Consultancy; Helocyte Inc: Research Funding; Kadmon: Consultancy. Al Malki:Miltenyi Biotec: Consultancy, Research Funding; Incyte: Consultancy, Research Funding; Hasna Biopharma: Membership on an entity's Board of Directors or advisory committees; NexImmune: Consultancy, Research Funding; Gilead: Consultancy, Research Funding; CareDx: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal